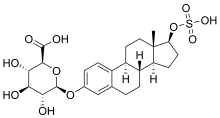
Estradiol 3-glucuronide 17β-sulfate
 | |
| Names | |
|---|---|
| IUPAC name 17β-(Sulfooxy)estra-1,3,5(10)-trien-3-yl β-D-glucopyranosiduronic acid | |
| Systematic IUPAC name (2S,3S,4S,5R,6S)-3,4,5-Trihydroxy-6-{[(1S,3aS,3bR,9bS,11aS)-11a-methyl-1-(sulfooxy)-2,3,3a,3b,4,5,9b,10,11,11a-decahydro-1H-cyclopenta[a]phenanthren-7-yl]oxy}oxane-2-carboxylic acid | |
| Other names E2-3G-17S | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI |
|
| ChemSpider |
|
| KEGG |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C24H32O11S |
| Molar mass | 528.57 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
Estradiol 3-glucuronide 17β-sulfate (E2-3G-17S) is an endogenous estrogen conjugate and metabolite of estradiol.[1][2] It is related to estradiol 3-sulfate and estradiol 17β-glucuronide.[1][2] Estradiol 3-glucuronide 17β-sulfate has 0.0001% of the relative binding affinity of estradiol for the ERα, one of the two estrogen receptors (ERs).[3] It shows less than one million-fold lower potency in activating the estrogen receptors relative to estradiol in vitro.[4]
See also
References
- ^ a b Isobe T, Shiraishi H, Yasuda M, Shinoda A, Suzuki H, Morita M (January 2003). "Determination of estrogens and their conjugates in water using solid-phase extraction followed by liquid chromatography-tandem mass spectrometry". J Chromatogr A. 984 (2): 195–202. doi:10.1016/s0021-9673(02)01851-4. PMID 12564690.
- ^ a b Katayama M, Matsuda Y, Shimokawa K, Kaneko S (June 2003). "Simultaneous determination of 16 estrogens, dehydroepiandrosterone and their glucuronide and sulfate conjugates in serum using sodium cholate micelle capillary electrophoresis". Biomed. Chromatogr. 17 (4): 263–7. doi:10.1002/bmc.236. PMID 12833391.
- ^ Durmaz V, Schmidt S, Sabri P, Piechotta C, Weber M (October 2013). "Hands-off linear interaction energy approach to binding mode and affinity estimation of estrogens". J Chem Inf Model. 53 (10): 2681–8. doi:10.1021/ci400392p. PMID 24063761.
- ^ Coldham NG, Dave M, Sivapathasundaram S, McDonnell DP, Connor C, Sauer MJ (July 1997). "Evaluation of a recombinant yeast cell estrogen screening assay". Environ. Health Perspect. 105 (7): 734–42. doi:10.1289/ehp.97105734. PMC 1470103. PMID 9294720.
- v
- t
- e
- Cholesterol
- 22R-Hydroxycholesterol
- 20α,22R-Dihydroxycholesterol
- Pregnenolone
- 11β-Hydroxypregnenolone
- 17α-Hydroxypregnenolone
- 21-Hydroxypregnenolone
- 17α,21-Dihydroxypregnenolone
- 11β,17α,21-Trihydroxypregnenolone
- Cholestanes: 24S-Hydroxycholesterol
- Cholesterol
- Pregnanes: 3α-Dihydroprogesterone
- 3β-Dihydroprogesterone
- 5α-Dihydrocorticosterone
- 5α-Dihydroprogesterone
- 5β-Dihydroprogesterone
- Allopregnanolone
- Corticosterone
- DHC
- DHDOC
- 11-Deoxycorticosterone
- Epipregnanolone
- Isopregnanolone
- Pregnanolone
- Pregnenolone
- Pregnenolone sulfate
- Progesterone
- THB
- THDOC
 | This article about a steroid is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











