Sibrafiban
Chemical compound
 | |
| Identifiers | |
|---|---|
| |
| CAS Number |
|
| PubChem CID |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) |
|
| Chemical and physical data | |
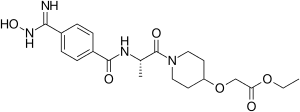
| Formula | C20H28N4O6 |
| Molar mass | 420.466 g·mol−1 |
| 3D model (JSmol) |
|
| |
InChI
| |
Sibrafiban (Ro 48–3657, proposed brand name Xubix) is the double prodrug of Ro-44-3888, which is a platelet aggregation inhibitor. It was being developed for secondary prevention of arterial thrombosis following unstable angina pectoris and acute myocardial infarction (MI).[1] On August 6, 1999, Hoffmann-La Roche announced that the preliminary results from Phase III clinical trials had not shown that sibrafiban was better than aspirin in preventing recurrent ischemic events in patients with acute coronary syndrome. The development of sibrafiban was terminated.
See also
References
- ^ Dooley M, Goa KL (February 1999). "Sibrafiban". Drugs. 57 (2): 225–30, discussion 231–2. doi:10.2165/00003495-199957020-00012. PMID 10188763. S2CID 263996772.
- v
- t
- e
| Glycoprotein IIb/IIIa inhibitors |
|
|---|---|
| ADP receptor/P2Y12 inhibitors | |
| Prostaglandin analogue (PGI2) | |
| COX inhibitors | |
| Thromboxane inhibitors | |
| Phosphodiesterase inhibitors | |
| Other |
| Vitamin K antagonists (inhibit II, VII, IX, X) | |||||
|---|---|---|---|---|---|
| Factor Xa inhibitors (with some II inhibition) |
| ||||
| Direct thrombin (IIa) inhibitors |
| ||||
| Other |
fibrinolytics
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
 | This drug article relating to the blood and blood forming organs is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











